Utilizing technology to modernize and develop new diagnostic and analytical devices
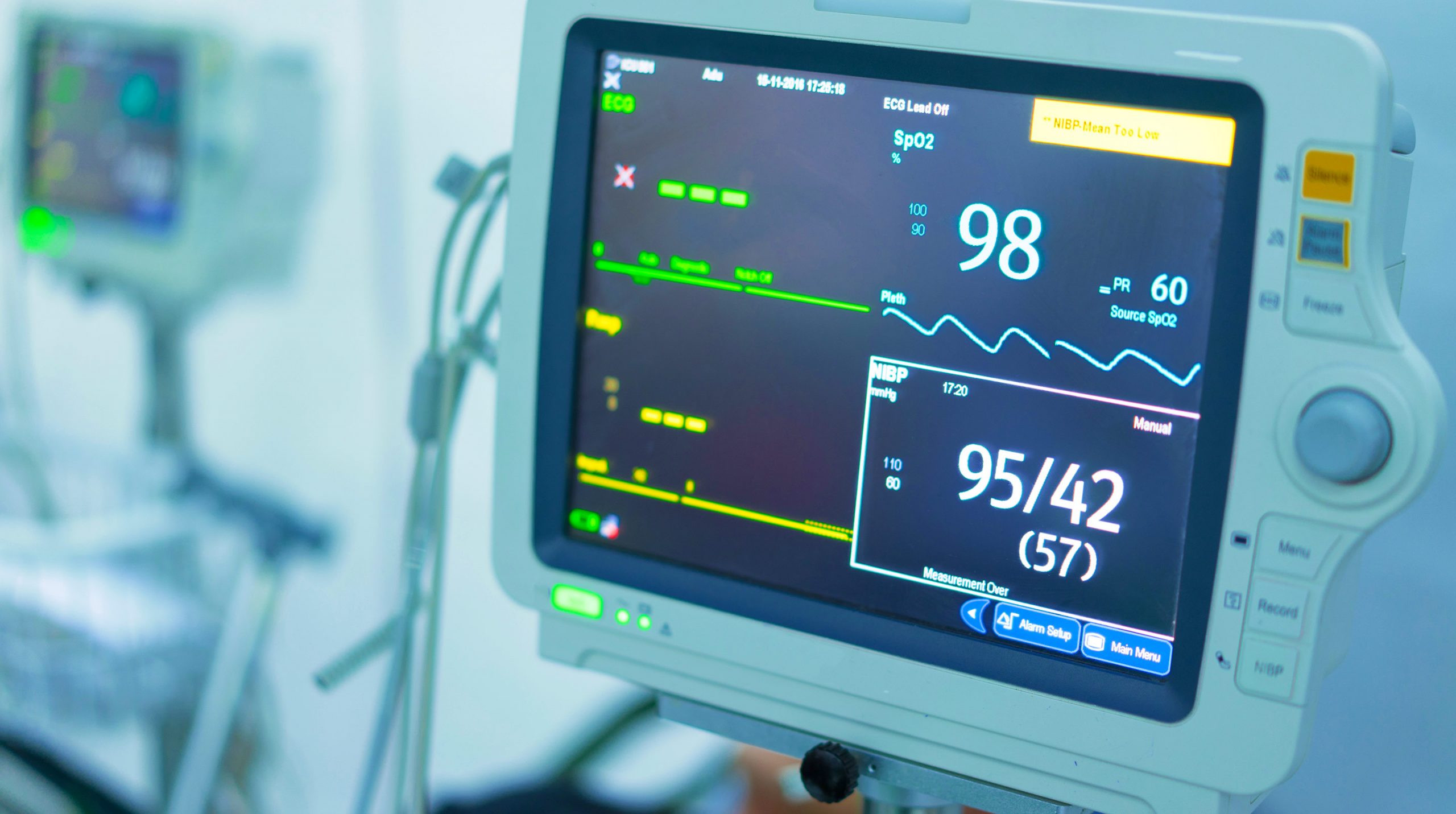
Diagnostics and Analytical Instruments are the front lines in the development and implementation of strategies for treatment, control, and in many cases prevention of disease and outbreaks. Their critical role in the health system has been brought into sharp focus by the current COVID-19 pandemic. However, one of the main challenges has been the need for manufacturers to develop products that enhance patient care, lower costs, and improve efficiency. Yet advances in mRNA, Software driven medical devices, Artificial Intelligence, Machine Learning, and robotics are changing the trajectory of the industry.
mRNA
mRNA has emerged as a software platform enabling not just COVID but personalized cancer vaccines. In addition to applications in infectious diseases, researchers in both academia and industry have been pursuing the use of mRNA vaccines to harness the immune system to fight cancer. This will beckon a new era for the application of mRNA towards infectious diseases, particularly as rapid response platforms to help deal with outbreaks.
Machine Learning, Artificial Intelligence & Robotics
Recent developments in robotics, artificial intelligence, and machine learning have put us on the cusp of a new automation age. AI is already helping improve process efficiency in the life sciences industry. The next three to five years are likely to prove AI’s value in R&D, especially in drug discovery, and across other areas of the value chain. Going forward, AI will enable major scientific breakthroughs, accelerating the creation of new therapies and vaccines to fight diseases.
Software-Driven Diagnostics and Medical Devices
Software-driven diagnostics medical devices gained magnifying importance with their ability to be independent of hardware and process accurate information for end-users. SaMDs are constantly evolving, posing opportunities and challenges for device organizations and regulators. The FDA recently acknowledged the prevalence of Artificial Intelligence/Machine Learning (AI/ML)-Based Software as Medical Devices (SaMDs) and has been taking steps towards advancing its regulatory oversight.
Join peers from leading Diagnostics and Analytical Instrument companies for an interactive executive salon exploring how emerging technologies are transforming our industry.

Utilizing technology to modernize and develop new diagnostic and analytical devices
Learn moreContact us
(*) Asterisk denotes mandatory fields






