“Finding the right clinical trial site is half the battle won.”
Despite decades of experience, clinical trial site selection still remains one of the biggest execution risks in clinical drug development. Patient recruitment stalls, project timelines slip and trial budgets balloon—not because therapies aren’t promising, but because the incorrect hospital sites get chosen during clinical trial operational planning.
The Ground Reality: Why Site Selection Is So Hard?
Across clinical trials and sponsors, common pain points recur:
- Recruitment & retention: Industry analyses repeatedly show ~85% of clinical trials struggle to recruit/retain to plan; 80%+ of delays tie back to enrolment issues; and ~37% of selected clinical sites miss targets, with some clinical sites enrolling zero patients.
- Time & cost blowouts: Many clinical trials double their original timelines to hit enrolment goals. Each day of delay can cost sponsors hundreds of thousands dollars in lost revenue and operating burn.
- Operational variability: Staffing shortages, data quality problems and infrastructure gaps (labs, calibration, SOPs) are unevenly distributed across clinical sites, making historical relationship-based selections risky.
What is the root cause?
Sponsors often rely on self-reported clinical site capabilities, anecdotal experience, or incomplete internal histories rather than triangulating multi‑source performance evidence and patient population signals at the clinical trial protocol level.
Reframing the Problem: Clinical Site Selection Framework
To choose the most suitable clinical trial site, an analyst should evaluate the options through these four lenses before making a decision.
- What is needed?
Protocol–to-Site Fit: A robust clinical site selection strategy involves not only evaluating site capabilities, but also ensuring that these capabilities align precisely with the protocol requirements. For this the clinical protocol document needs to be translated to a “Site capability fingerprint” allowing precise clinical site selection based on the defined trial requirements. - Who has delivered?
Investigator & Clinical Site Performance: Once the list of experienced sites meeting protocol requirements has been collated, the next step is to examine actual enrolment rates, startup times, screen failures, retention and deviations, to arrive at final list of top-performing sites most suited to the clinical trial’s needs. - Where are the patients?
Patient Availability: With enrolment rate hindering the success of most of the sites, it’s imperative to have confidence in patient availability of the selected site locations. In order to do so, analysts have to leverage claims/EHR derived Real World Data (RWD) to geospatially estimate eligible patient pools (by diagnosis codes, treatments, comorbidities, age/sex distributions). - What is the regulatory onus?
Risk mitigation: Finally, ensure that chosen sites not only meet operational needs but also uphold the highest standards of quality and ethics. Tapping into Regulatory inspection reports, audit findings, deviation logs and warning letters from authorities like FDA, EMA, MHRA, etc. can help prioritize sites with strong regulatory track records.
Put together, these four lenses form an evidence-based site checklist, ranking sites by expected enrolment velocity, data integrity, operational resilience and regulatory compliance for the clinical trial protocol at hand.
The Data Universe: What to Use and Who Has It?
Before exploring any AI-driven solution for business challenges, it is crucial to assess the data availability. Here is a list of noteworthy data sources that sponsors or Clinical Research organizations (CROs) must leverage, as they decide to bolster their operations with AI.
A) Commercial Intelligence
The industry has now evolved to offer availability of consortia based global data repository with multisource investigator/site performance metrics (enrolment, startup), offering vast curated commercially available clinical trial datasets that can enable data backed feasibility beyond one sponsor’s history.
B) Internal Operational Data
Sponsors or CROs have been running trials for decades, continually updating their data repositories. Many are now seeking to modernize clinical repositories to better leverage data from internal sources like CTMS/EDC/eTMF/EDW consolidating trial and site specific—startup cycle times, screen failures, enrolment cadence, deviations, monitor findings, query resolution times.
C) Patient Level Signals
Administrative claims data are an important real world data source for pragmatic clinical trials because they offer longitudinal information on demographics and reimbursed healthcare services, that can help to estimate local eligible patient density and care seeking patterns relevant to the protocol. ‑world data source for pragmatic clinical trials because they offer longitudinal information on demographics and reimbursed healthcare services,
D) Publicly available Clinical Research Databases Publicly available clinical research databases, such as ClinicalTrials.gov, EudraCT, and WHO ICTRP, provide open access to global trial records and investigator profiles. These resources enable sponsors and CROs to find experienced sites/investigators and validate feasibility using real-world evidence beyond proprietary datasets.
(AI)InSite: From AI Insights to Decisions
Persistent Systems has developed (AI)InSite, an advanced, machine learning and agentic AI-powered platform designed to transform clinical site selection from a manual, intuition-driven process into a data-driven, evidence-based workflow. By leveraging clinical trial protocol analysis, curated data retrieval and AI-powered scenario ranking, (AI)InSite unlocks smart clinical site selection for sponsors and CROs.
From our (AI)Insite pilots and adjacent client programs, we consistently see:
- Faster feasibility: AI‑assisted shortlists in minutes with curated internal and external sources.
- Improved enrolment velocity: Better protocol–to-site fit yields.
- Site performance and reduced risk: Early visibility into performance metrices, data quality and staffing risks.
To bring (AI)InSite function in motion, large pharma companies designed a step-wise approach that acts as an execution blueprint—progressing from understanding current processes to defining ROI, piloting, iterating, scaling, and continuously refining the system. Here’s a quick glance:
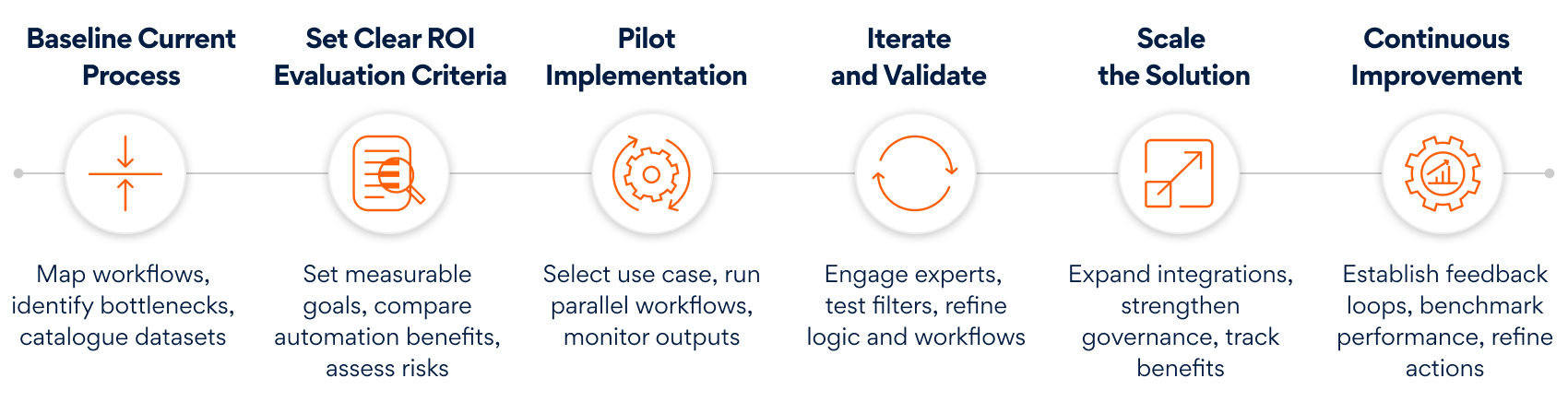
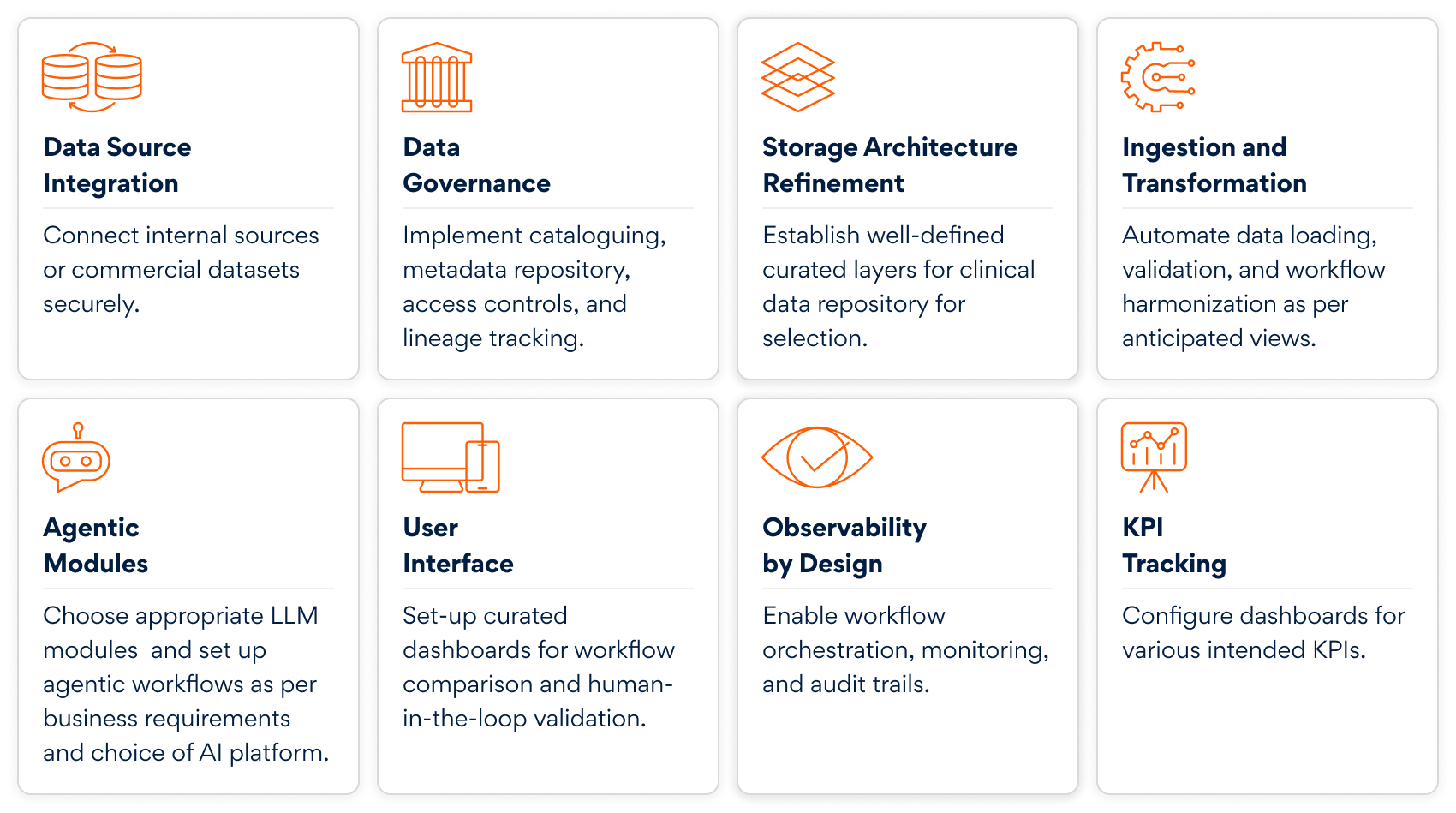
Key technical steps are needed for (AI)InSite setup to execute pilot projects:
In summary, if typical clinical trial feasibility conversations start with “Who do we know in Country X?”, it is now time to start with “Where are the patients?”, “What does the protocol truly demand?”, “Which sites have delivered for these demands?”, and “What is the regulatory onus?”, all before the CRO sends the first feasibility questionnaire.
With Persistent Systems (AI)InSite accelerator, platform engineering expertise and strong domain expertise, Sponsors and CROs can unlock intelligent, secure and scalable clinical site selection workflows.
The future of AI-powered clinical research is here; let’s build it together. To know more, get in touch with us here.













